The Results are In: Key Takeaways from 2017 eCQM Reporting

Today marks the last day to submit your eCQM files to the CMS Inpatient Quality Reporting (IQR) program. How did you do? Did you breeze through year two of the eCQM requirement or was it a struggle for your hospital?
And while The Joint Commission is giving hospitals a bit more time for eCQM submission, the summer deadline is quickly approaching. Have you crossed this submission off of your 2018 to-do list yet?
Now that all of our hospitals have completed submissions for the CMS IQR program, and nearly all of them have completed their submissions for The Joint Commission, we wanted to share some of the results with you. Drum roll please!
Submission Process
Let’s review the actual CMS IQR submission process. Keep in mind that as a vendor, our process looks a little different than, say, if you were a hospital that was submitting to CMS on your own.
Below are the general steps that we take with each client.
- Prep: Medisolv works with clients throughout the reporting year to help the client identify what data to submit (which measures from which quarter). Once a client authorizes Medisolv as a submission vendor with QualityNet, they can generate their EHR Certification ID with CHPL.
- Snapshot: To take a snapshot of their data, clients can click the data submission snapshot button in our ENCOR software, which saves a copy of all eCQM data for the selected measures and reporting period. This data is then used for the QRDA file generation and submission to the various agencies, future data review and audit purposes if needed. After sending a snapshot, the client completes the Medisolv approval process, and the submission team receives an email for internal approval and production submission authorization.
- Test: In addition to internal testing, Medisolv begins testing select client data by using the CMS testing environment.
- Resolve: Medisolv reviews the test files and works on resolving any errors we find before beginning the actual process of submission.
- Submit: Once the submission window opens up, Medisolv submits the files (now called production files) to CMS.
- Resolve Again: Medisolv reviews the results of the individual client's submission and begins the process of resolving any additional errors. Sometimes the error is a technical error. In that case, Medisolv figures out where the error is and then applies the fix. Sometimes the error is a data error. When it’s a data error, we work with the client to help identify what went wrong on the client side and help them to fix the issues.
- Resubmit If Necessary: After all errors have been resolved, the client then takes a new snapshot and Medisolv resubmits the file to CMS.
- Complete: This process continues until the files comes back as completely clean.
Submission Timeline
CMS and The Joint Commission have different timelines and deadlines for submission. For instance, in 2017, CMS opened their portal for 2017 data submissions by May. The Joint Commission, on the other hand, said they would open their portal by October of 2017 but were delayed until January of 2018.
Either way, Medisolv begins testing and submissions as soon as possible to give ourselves room for resolving errors.
For CMS submissions, Medisolv began testing files in the spring of 2017, and by August of 2017, we had our first completed submission. The majority of our clients were submitted by October of 2017, but we still had a few pending submissions that we finished up by the end of February, 2018. CMS recently extended the deadline to Friday, March 16.
With The Joint Commission, the process was longer. Despite the fact that their actual submission portal didn’t open in October as planned, they did open the testing portal. Medisolv started testing files in October of 2017. Once the portal opened up in January of 2018, we began our submission process and had our first completed submission by mid-January.
Even though the majority of our clients have completed their eCQM submissions to The Joint Commission, we still have some hospitals left to submit before the June 29, 2018 deadline.
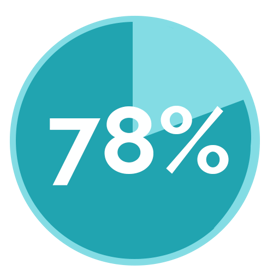
Submission Stats
Percentage of Hospitals Submitted
| CMS IQR Program | The Joint Commission ORYX® |
 |
 |
Total Number of Files Submitted
| All Programs |
 |
Average Number of Measures Submitted Per Hospital

*Only four measures are required to be submitted for either program.
Percent of hospitals submitting over the required number of measures
| All programs |
 |
Top Four Submitted eCQMs
| CMS |
|
| 2. STK-2 (56% of clients submitted this measure) |
| 3. STK-5 (52% of clients submitted this measure) |
| 4. VTE-2 (50% of clients submitted this measure) |
| The Joint Commission |
|
| 2. STK-2 (71% of clients submitted this measure) |
| 3. VTE-2 (69% of clients submitted this measure) |
| 4. STK-5 (64% of clients submitted this measure) |
And what are we most proud of? The significant improvement in eCQM performance. A lot of time, resources, effort and PATIENCE went into getting these rates to where they are today. Most of our hospitals are able to use the eCQM data not just to improve performance but also to improve the quality of care they are delivering to their patients.
How does your eCQM data compare?
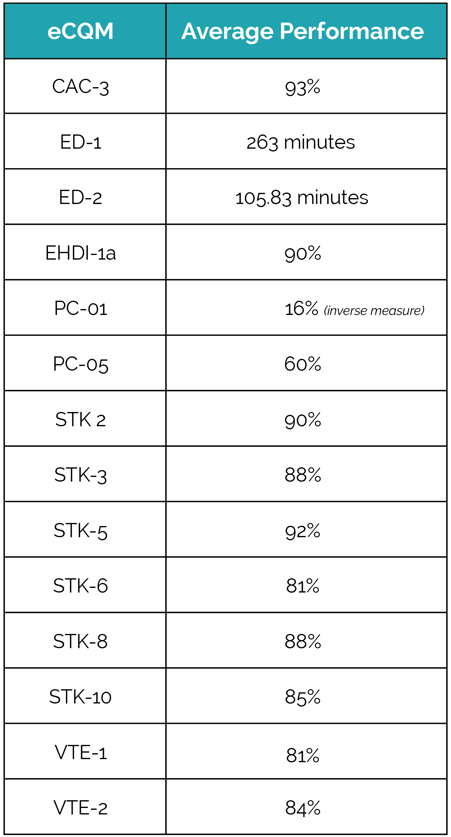
Big shout out to our clients who worked so hard to meet their eCQM requirements for CMS and The Joint Commission this year.
If you are ready to conquer your eCQM submissions, talk to Medisolv today. We have successfully submitted eCQM data to CMS on our client’s behalf since 2012. Feel good about your submissions and the accuracy of your eCQM data with Medisolv.
Get Ahead and Improve Your QualityMedisolv Can Help We've seen some big changes in the CMS eCQMs. With Medisolv as your partner we help guide you through every new change along the way. Talk with us about how we can help you track and improve your quality performance. Blog Post: "Survey Results: The eCQM Process" |





Comments